Use and Important Safety Information
What is
the most important safety
information I should know about RAVICTI?
RAVICTI may cause serious side
effects,
including:
Nervous
system side
effects
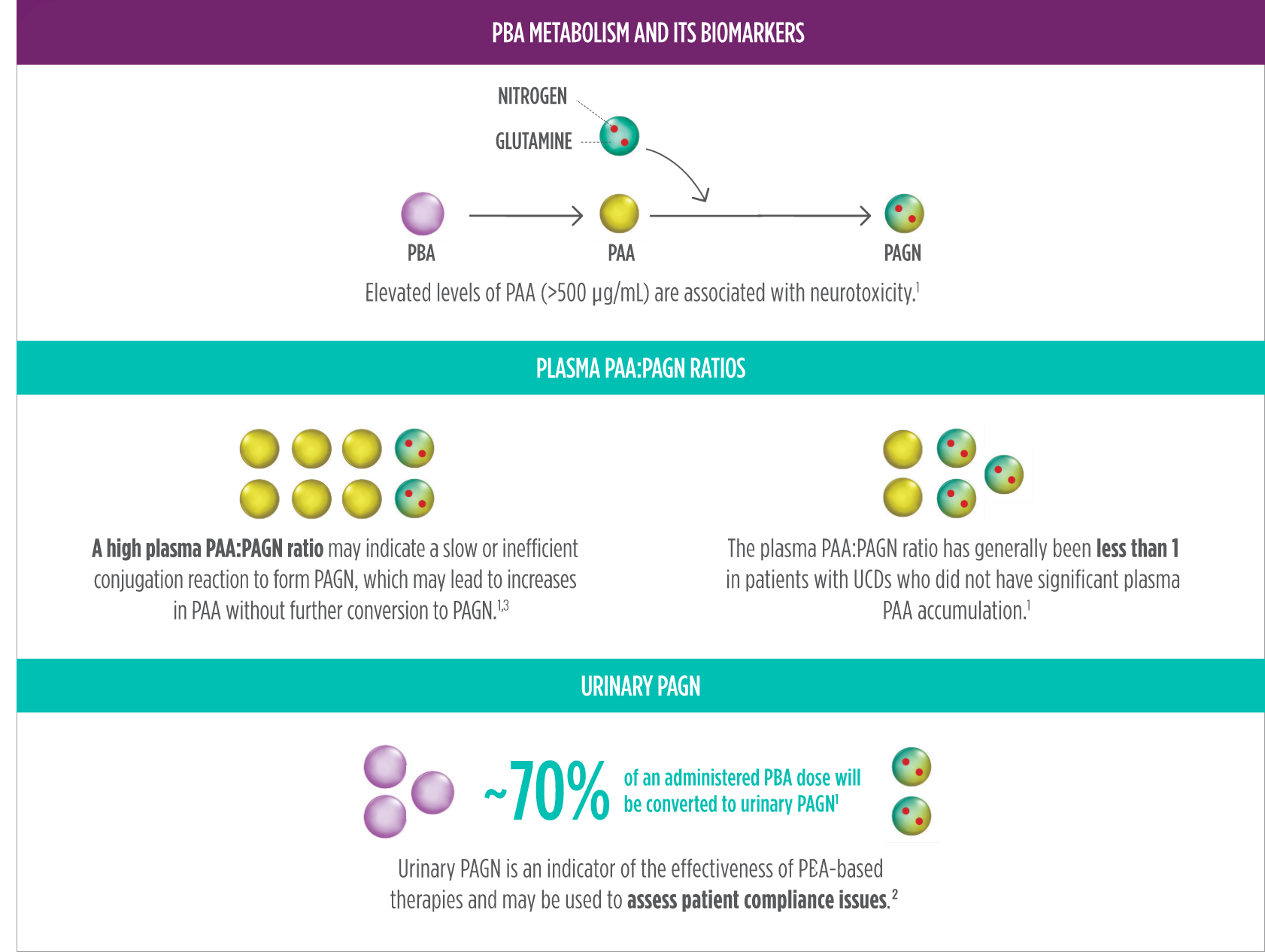
(Neurotoxicity) Phenylacetate (PAA),
a
breakdown product of RAVICTI, may cause nervous system side effects.
Call
your doctor or get medical help right away if you have any of these
symptoms while taking RAVICTI:
- sleepiness
- lightheadedness
- change in taste
- problems with hearing
- confusion
- problems with memory
- worsening of numbness, tingling, or burning in your hands or feet
- headache
- feeling very tired (fatigue)
- nausea
- vomiting
Your doctor may do blood tests to measure the amount of PAA in your blood during your treatment with RAVICTI.
What is RAVICTI?
- RAVICTI (glycerol phenylbutyrate) Oral Liquid is a prescription medicine used for long-term management of high blood levels of ammonia (hyperammonemia) caused by a condition called a urea cycle disorder (UCD). RAVICTI should be used if the UCD cannot be managed with a low-protein diet and dietary supplements alone. RAVICTI must be used along with a low-protein diet and in some cases dietary supplements.
- RAVICTI is not used for the acute treatment of hyperammonemia in people with UCD.
- It is not known if RAVICTI is safe and effective for the treatment of N-acetylglutamate synthase (NAGS) deficiency.
Do not take RAVICTI if you are allergic to phenylbutyrate. Call your doctor or go to the nearest hospital emergency room if you have wheezing, shortness of breath, cough, low blood pressure, flushing, nausea or a rash while taking RAVICTI.
Before taking RAVICTI, tell your doctor about all of your medical conditions, including if you:
- have liver or kidney problems.
- have pancreas or bowel (intestine) problems.
- are pregnant or plan to become pregnant. It is not known if RAVICTI will harm your unborn baby. If you become pregnant during treatment with RAVICTI, call Amgen at 1‐866‐479‐6742 to report the pregnancy.
- are breastfeeding or plan to breastfeed. It is not known if RAVICTI passes into your breast milk. Breastfeeding is not recommended during treatment with RAVICTI. Talk to your doctor about the best way to feed your baby if you take RAVICTI.
What are possible side effects of RAVICTI?
RAVICTI may cause serious side effects, including:
-
See "What is the most important information I should know about RAVICTI?"
The most common side effects of RAVICTI in adults include:
- diarrhea
- gas
- headache
- abdomen (stomach) pain
- vomiting
- tiredness
- decreased appetite
- indigestion or heartburn
The most common side effects of RAVICTI in children 2 years to 17 years of age include:
- upper abdomen (stomach) pain
- rash
- nausea
- vomiting
- diarrhea
- decreased appetite
- headache
The most common side effects of RAVICTI in children 2 months to less than 2 years of age include:
- low white blood cell count (neutropenia)
- vomiting
- constipation
- diarrhea
- fever
- reduced food intake
- cough
- stuffy nose
- runny nose
- skin rash
- small round bumps on the skin
The most common side effects of RAVICTI in children less than 2 months of age include:
- vomiting
- rash
- gastroesophageal reflux
- increased levels of liver enzymes in the blood
- decreased appetite and reduced food intake
- low red blood cell count (anemia)
- cough
- loss of too much body fluid (dehydration)
- too much acid in the blood (acidosis)
- high blood platelet count (thrombocytosis)
- low blood platelet count (thrombocytopenia)
- low blood neutrophil count (type of white blood cell) (neutropenia)
- high white blood cell count (lymphocytosis)
- diarrhea
- gas
- constipation
- fever
- drowsiness (lethargy)
- irritability
- agitation
These are not all of the possible side effects of RAVICTI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
For additional Important Safety Information, click here for the Medication Guide and discuss with your doctor.
Use and Important Safety Information
What is
the most important safety
information I should know about BUPHENYL?
BUPHENYL may cause serious side
effects,
including:
Nervous
system side
effects
(Neurotoxicity) Phenylacetate (PAA),
a
breakdown product of BUPHENYL, may cause nervous system side
effects. Call
your doctor or get medical help right away if you have any of these
symptoms while taking BUPHENYL:
- sleepiness
- weakness
- lightheadedness
- problems with memory
- worsening of numbness, tingling, or burning in your hands or feet
- change in taste
- problems with hearing
- confusion
- headache
What is BUPHENYL?
- BUPHENYL® (sodium phenylbutyrate) Tablets is a prescription medicine that can be taken by mouth and BUPHENYL® (sodium phenylbutyrate) Powder is a prescription medicine that can be taken by mouth or feeding tube for the long-term management of high blood levels of ammonia (hyperammonemia) caused by a condition called a urea cycle disorder (UCD).
- BUPHENYL only treats high blood levels of ammonia in patients with carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC) and argininosuccinic acid synthetase (AS) deficiencies.
- BUPHENYL can be used in infants up to 28 days old who have a complete enzyme deficiency, and in patients 1 month of age and older who have a partial enzyme deficiency and have a history of brain damage from high blood levels of ammonia.
- BUPHENYL must be used along with a low-protein diet and in some cases, dietary supplements.
- BUPHENYL is not used to treat acute (severe) hyperammonemia, which is a medical emergency.
Do not take BUPHENYL if you are allergic to phenylbutyrate. Call your doctor or go to the nearest hospital emergency room if you experience an allergic reaction while taking BUPHENYL.
Before taking BUPHENYL, tell your doctor about all of your medical conditions, including if you:
- have heart failure or decreased kidney function, which may lead to retention of the sodium content of BUPHENYL with potentially serious consequences, such as worsening heart failure, high blood pressure, and swelling.
- are pregnant or plan to become pregnant. It is not known if BUPHENYL will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if BUPHENYL passes into your breast milk. Breastfeeding is not recommended during treatment with BUPHENYL. Talk to your doctor about the best way to feed your baby if you take BUPHENYL.
What are possible side effects of BUPHENYL?
BUPHENYL may cause serious side effects, including:
-
See “What is the most important information I should know about BUPHENYL?”
The most common side effects of BUPHENYL include:
- decreased appetite
- body odor
- bad taste
- absent or irregular periods in women
These are not all of the possible side effects of BUPHENYL. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
For additional Important Safety Information, click here for the Patient Package Insert and discuss with your doctor.