Provider Support
A representative can answer questions and provide more information about RAVICTI.
Review dosing and administration information for your patient starting on or switching to RAVICTI
This tool is intended to provide dose calculations based on patient body surface area (BSA). For additional information about dosing and administration, please refer to the Full Prescribing Information.
1. Select the age range of your patient.
2. Enter the height and weight of your patient in desired units to determine BSA.a The BSA is then used to calculate the daily dosage range.b
3. Drag the slider below to select the daily dosage from the calculated range.c Be sure to follow the guidelines outlined in Need to Know.
4. Select number of doses per day.
This is the dose of RAVICTI for your patient, which includes proper rounding.
Multiply this dose by 90 in order to find your patient’s prescription amount for 3 doses per day for 30 days.
The results may be printed or emailed once calculated. Note the email address provided will not be registered for educational or promotional emails and is used solely for delivering the custom dosing results.
1. Choose the form of sodium phenylbutyrate (NaBPA).
2. Select the age range of your patient.
3. Enter the current daily dosage of NaBPA.d,e
4.Select number of RAVICTI doses per day.
This is the dose of RAVICTI for your patient, which includes proper rounding.
Multiply this dose by 90 in order to find your patient’s prescription amount for 3 doses per day for 30 days.
The results may be printed or emailed once calculated. Note the email address provided will not be registered for educational or promotional emails and is used solely for delivering the custom dosing results.
aBSA can be calculated by multiple equations; the Mosteller equation is used on this website.2,3
bThe recommended dosage range, based on BSA, in patients naïve to phenylbutyrate is 4.5 to 11.2 mL/m2/day (5 to 12.4 g/m2/day). For patients with some residual enzyme activity who are not adequately controlled with protein restriction, the recommended starting dosage is 4.5 mL/m2/day. These values are multiplied by your patient’s BSA to calculate the daily dosage range indicated in the slider.1
cThe maximum total daily dosage of RAVICTI is 17.5 mL (19 g); any dose value calculated on this website cannot exceed this value.1
dThe total daily dosage of RAVICTI (mL) equals the total daily dosage of NaPBA tablets (g) x 0.86.1
eThe total daily dosage of RAVICTI (mL) equals the total daily dosage of NaPBA powder (g) x 0.81.1
A representative can answer questions and provide more information about RAVICTI.
Determine your patient’s plasma PAA:PAGN ratio and urinary PAGN level.
The recommended dosage range, based on BSA, in patients naïve to phenylbutyrate is 4.5 to 11.2 mL/m²/day (5 to 12.4 g/m²/day).1
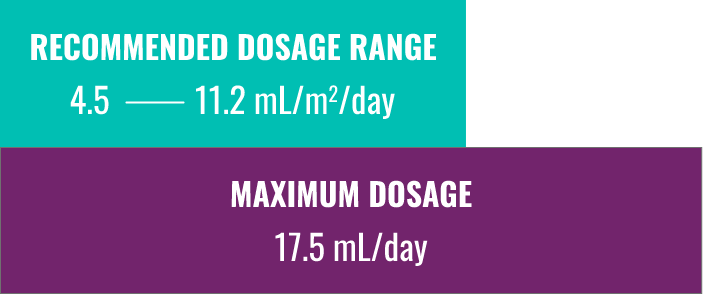
For patients with some residual enzyme activity who are not adequately controlled with protein restriction, the recommended starting dosage is 4.5 mL/m²/day.1
For patients with moderate to severe hepatic impairment, the recommended starting dosage is at the lower end of the recommended range (4.5 mL/m²/day) and should be kept at the lowest dosage necessary to control the patient’s plasma ammonia.1
An initial estimated RAVICTI dose for a 24-hour period is 0.6 mL RAVICTI per gram of dietary protein ingested.1
Poor adherence with prescribed diets will increase your patient’s protein intake, which may necessitate increasing dosage.1
Patients 2 years of age and older: give RAVICTI in 3 equally divided doses, each rounded up to the nearest 0.5 mL.1
Patients less than 2 years of age: give RAVICTI in 3 or more equally divided doses, each rounded up to the nearest 0.1 mL.1
Please note: the RAVICTI dose is automatically rounded up when using the calculator.
The maximum total daily dosage is 17.5 mL (19 g).1 Please note: the dosage calculator will not exceed the maximum.
RAVICTI must be used with dietary protein restriction and, in some cases, dietary supplements (e.g., essential amino acids, arginine, citrulline, protein-free calorie supplements).1
fOf the 51 adult patients in the 12-month study of RAVICTI, 7 patients (14%) reported a total of 10 hyperammonemic crises. Of the 26 pediatric patients 6 to 17 years of age in both 12-month studies of RAVICTI, 5 patients (19%) reported a total of 5 hyperammonemic crises. Of the 17 pediatric patients 2 months to less than 2 years of age in 3 open-label studies, 7 patients (41%) reported a total of 11 hyperammonemic crises. Of the 16 pediatric patients less than 2 months of age in an uncontrolled, open-label study, 5 patients (31%) reported a total of 7 hyperammonemic crises.1
gFor patients who require a volume of less than 1 mL per dose via nasogastric or gastrostomy tube, the delivered dose may be less than anticipated. Closely monitor these patients using ammonia levels.1
Download the Dosing Brochure to learn about dosing and administration of RAVICTI.
This assessment tool can help evaluate treatment of your patient and ensure management of their UCD.
View videos with your patient to help ensure correct administration of RAVICTI.
References: 1. RAVICTI (glycerol phenylbutyrate) Oral Liquid [prescribing information] Horizon. 2. Mosteller RD. Simplified calculation of body-surface area. N Engl J Med. 1987;317(17):1098. 3. Verbraecken J, Van de Heyning P, De Backer W, Van Gaal L. Body surface area in normal-weight, overweight, and obese adults: a comparison study. Metab Clin Exp. 2006;55(4):515-524. 4. Diaz GA, Krivitzky LS, Mokhtarani M, et al. Ammonia control and neurocognitive outcome among urea cycle disorder patients treated with glycerol phenylbutyrate. Hepatology. 2013;57(6):2171-2179.
RAVICTI (glycerol phenylbutyrate) Oral Liquid is indicated for use as a nitrogen-binding agent for chronic management of patients with urea cycle disorders (UCDs) who cannot be managed by dietary protein restriction and/or supplementation alone. RAVICTI must be used with dietary protein restriction and, in some cases, dietary supplements (e.g. essential amino acids, arginine, citrulline, protein-free calorie supplements).
The most common adverse reactions reported in clinical trials (at least 10% of patients) were:
Please see Full Prescribing Information.
RAVICTI (glycerol phenylbutyrate) Oral Liquid is indicated for use as a nitrogen-binding agent for chronic management of patients with urea cycle disorders (UCDs) who cannot be managed by dietary protein restriction and/or supplementation alone. RAVICTI must be used with dietary protein restriction and, in some cases, dietary supplements (e.g. essential amino acids, arginine, citrulline, protein-free calorie supplements).
The most common adverse reactions reported in clinical trials (at least 10% of patients) were:
Please see Full Prescribing Information.